hVIVO
Core Solutions
hVIVO is a purpose-built, full-service clinical development partner, integrating clinical trial delivery, specialist laboratories, consultancy, and world-leading human challenge trial services to generate decisive human data - fast.
Working with global biopharma and emerging biotech companies, we provide an accelerated, de-risked pathway to clinical proof-of-concept. Our end-to-end model connects expert study design, rapid recruitment, purpose-built facilities, and advanced laboratory capabilities to deliver high-quality, actionable insights with speed and precision.
From early strategy through to clinical execution and data delivery, hVIVO enables smarter development decisions, helping bring vital medicines to patients faster.
We are proud to announce that the companies within the hVIVO Group, hVIVO, Cryostore, CRS and Venn Life Sciences - will now operate under a single brand: hVIVO.
Find Out MoreFull-Service
Early Phase CRO
To support rapid study start-up and reliable delivery, our dedicated recruitment teams in Germany and the UK provide direct access to both healthy volunteers and patient populations. This is complemented by our integrated drug development consultancy as well as our infectious disease and immunology laboratories and biobanking services.
100+
Years of Service, Reputation & Trust
Over a century of combined experience in clinical research and infectious disease. Our heritage underpins the expertise and credibility we bring to every study.
5
Key Areas of Expertise
Integrated capabilities across consultancy, clinical trials, laboratories, human challenge studies, and recruitment. Together, these services provide a seamless end-to-end development platform.
400k
Trial Participants Database
Access to one of the largest specialist volunteer databases in Europe. This enables rapid recruitment and high retention to keep studies on track.
1.9k
Trials Completed & Counting
A proven track record of successfully delivering a high volume of clinical trials across multiple phases and therapeutic areas. We focus on quality, efficiency, and reliable outcomes.
5
Clinical Sites
hVIVO Integrated Service Platform
Your clinical development partner
hVIVO is a full-service, early-phase clinical development partner and the global leader in human challenge trials. With integrated capabilities spanning consultancy, clinical trials, laboratory services, recruitment, and storage, the Company supports biopharma clients in accelerating development timelines and generating high-quality data. Its purpose-built facilities and expert-led approach enable efficient, end-to-end delivery across complex therapeutic areas.

Delivering trials across all phases
hVIVO delivers clinical trial services across Phase I to Phase III, supporting the progression of therapies from first-in-human studies through to later-stage development. With specialist expertise in early-phase and complex study designs, the Company provides efficient trial execution, high-quality data, and seamless integration with laboratory and recruitment capabilities to optimise outcomes and accelerate development programmes.

Strategic guidance at every stage
hVIVO’s consultancy services provide strategic and scientific guidance across the clinical development lifecycle. From regulatory strategy and trial design to due diligence and investor readiness, the team supports clients in making informed decisions at every stage. Combining deep therapeutic expertise with practical delivery experience, hVIVO helps optimise development pathways and reduce risk.

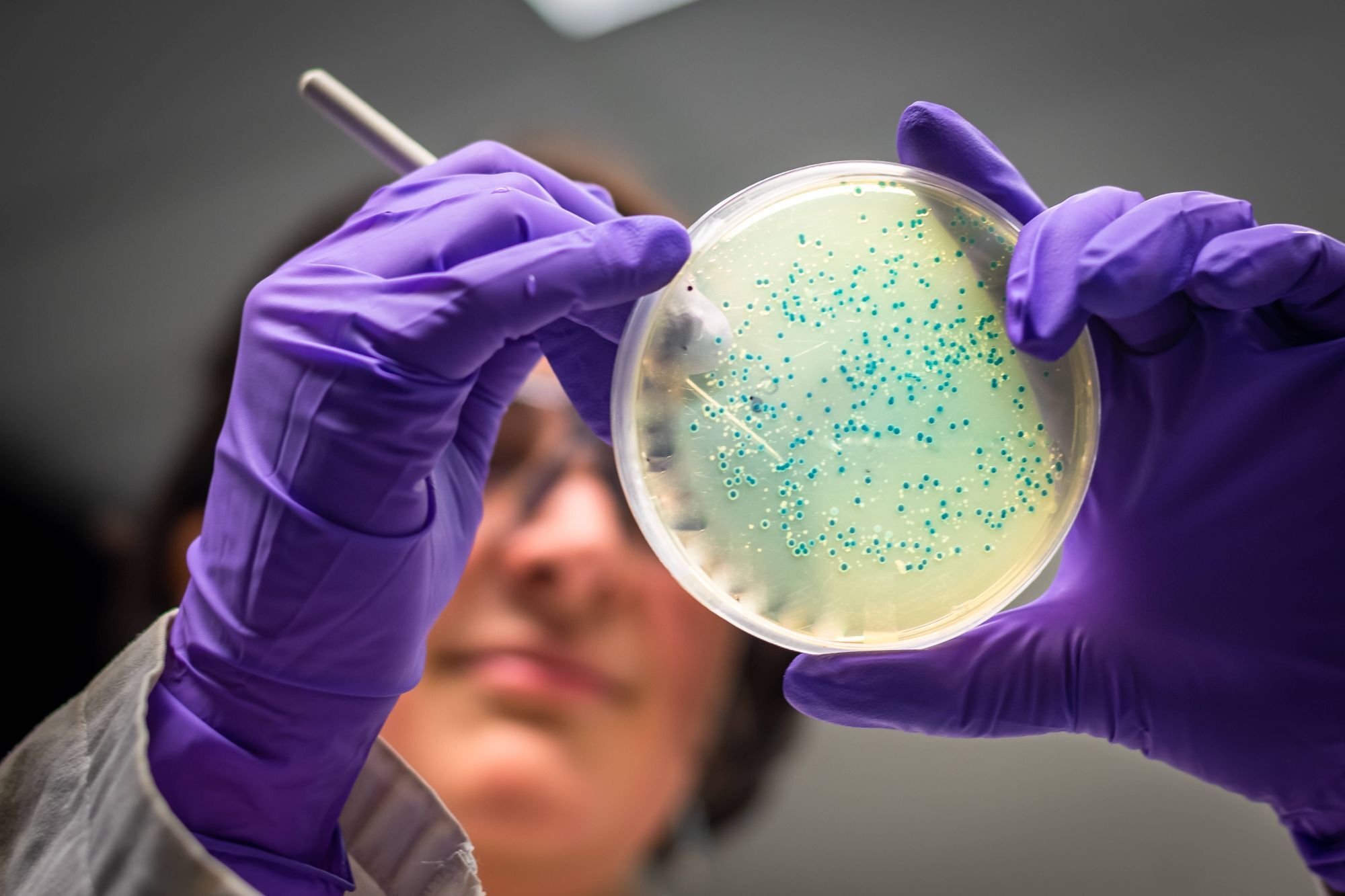
Advanced laboratory capabilities
hVIVO offers comprehensive laboratory services to support clinical trials and translational research, including bioanalytical testing, virology, immunology, and biomarker analysis. Its state-of-the-art facilities, including CL3 laboratories, enable high-quality sample processing and data generation. Fully integrated with clinical operations, these capabilities ensure consistency, speed, and scientific rigour across studies.

Decades of experience
hVIVO is the global leader in human challenge trials, with a heritage dating back decades and unmatched operational expertise. These studies involve the controlled exposure of volunteers to pathogens to generate early efficacy data, enabling faster and more cost-effective decision-making. hVIVO’s purpose-built quarantine facilities and specialist models support a wide range of infectious diseases.

Secure and compliant storage
hVIVO provides secure, compliant storage solutions for biological samples and clinical materials through its specialist cryostorage capabilities. With robust infrastructure and quality-controlled processes, the Company ensures the integrity and traceability of samples across the clinical development lifecycle. These services are fully integrated with laboratory and clinical operations for seamless handling and access.

Industry-leading recruitment

Sectors we service
Accelerating Clinical Development Through Human Challenge Trials
By working with pharmaceutical, biotechnology, and emerging companies, we help accelerate development timelines, reduce risk, and support confident decision-making at critical milestones. Our commitment to scientific rigour, operational excellence, and innovation ensures we remain at the forefront of human challenge studies and clinical research globally.
Need comprehensive expertise and support to accelerate drug development?
Find out how hVIVO can help you.
Investors
hVIVO is a publicly traded company on the London Stock Exchange AIM Market under the symbol HVO.
Participants
hVIVO is currently recruiting participants to take part in our clinical trials to further medical research.