If there would be a survey of 100 clinical research professionals either from CRO or from Pharma/Biotech industry to explain what a human challenge study is, the most common answer most probably would be – “I have never heard of it” and only very few of them would give the correct description. This is not something extraordinary, as this type of clinical studies is unique and niche, and highly therapeutically focused. A different situation in responses would be if the respondents of such survey would be those working in the field of infectious diseases or early clinical development of vaccines.
Even though human challenge studies or controlled human infection model (CHIM) studies may sound like something new within the clinical research space, over the past 80 years they have been conducted for over 20 different infectious disease pathogens and over 45,000 volunteers have participated in these clinical trials (1,2). Over 200 CHIM studies have been registered on clinicaltrials.gov. The numbers will definitely increase in the years to come, especially due to significantly increased interest in vaccine and antiviral development after the COVID-19 pandemic (3). This article overviews human challenge studies, specific aspects of their conduct and safety for the healthy volunteers participating in CHIM studies.
Human challenge studies or CHIM studies is a specific type of clinical trials, which involve the intentional infection of a healthy, adult, consenting volunteers with an infectious agent (3). In other words, healthy volunteers enrolled in these studies are infected on purpose with a particular pathogen, in order to investigate the efficacy of vaccines, antivirals, anti-infectives or anti-parasitic drugs.
Even though infecting a healthy person on purpose might sound very unethical or even dangerous, these studies are conducted in a highly controlled environment. Moreover, there is a proven track record of safety of these clinical trials discussed hereinafter in this article.
Even though human challenge studies may sound as very complex and complicated, from their set-up and management perspective many elements are very similar or even the same as for any other clinical trial. On the other hand, pathogen inoculation and disease control require specific experience and environment, therefore, these studies are mostly conducted in specialized units.
From a regulatory perspective, CHIM studies are considered to be either Phase Ib, Phase I/II or Phase IIa clinical trials, therefore, the same study start-up timelines apply. Usually, first in human data have to be obtained prior to the conduct of human challenge study. The flow of a human challenge study is shown in Figure 1. Some specific aspects of healthy volunteer screening and consent, randomization and treatment, as well as monitoring and follow-up are discussed further.
Figure 1. Flowchart of a human challenge study
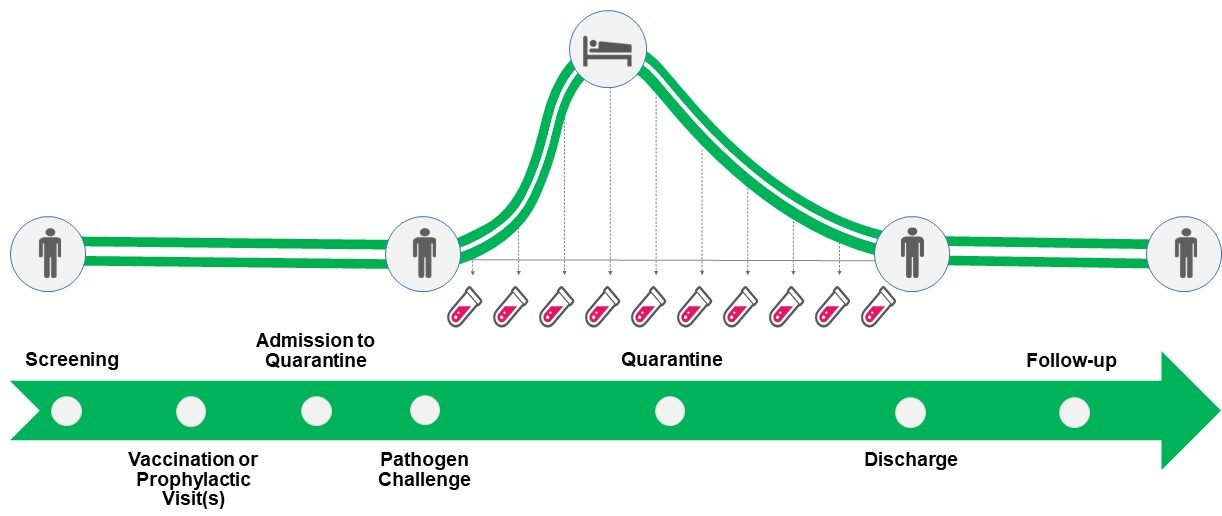
Controlled human infection model studies, as any other healthy volunteer clinical trials starts with healthy volunteer screening. Depending on the pathogen used in a challenge study, many healthy volunteers are not serologically suitable to participate in the study, due to the high immunity against the pathogen and therefore, low probability to get infected. This means that the number of subjects screened for a challenge study might be much higher in comparison to the usual healthy volunteer clinical trial.
The ethical aspects of CHIM clinical trials have been widely debated, with specific focus on the informed consent process and the understanding of risks and benefit (4,5). In order to be included in a challenge study, healthy volunteers are thoroughly screened based on inclusion and exclusion criteria and enrolled after an informed consent process. Informed consent form is usually quite extensive, as it must include not only the information related to investigational medicinal product, but also to the pathogen inoculation and associated risks of the infection. Therefore, informed consent process may frequently include a test of understanding (3).
The selected study participants are randomized to receive either the investigational medicinal product (IMP), comparator (if applicable) or placebo, and at a certain timepoint challenged with the infective pathogen. Worth mentioning that not all study participants may get infected with the disease after the pathogen has been inoculated, some may experience asymptomatic disease.
Based on the type of IMP (vaccine, therapeutic or prophylaxis), timing of the dosing and challenge pathogen inoculation may differ. While vaccines are usually administered 1 or 2 months prior to the challenge, prophylactic or therapeutic treatments may be administered just before or after pathogen inoculation, based on the treatment regimen and the onset of disease. Depending on the pathogen, participants may be treated with rescue medication at a defined time based on the study protocol and development or severity of symptoms.
Challenge studies requires careful monitoring throughout the clinical trial period, vital signs, and other clinical and laboratory parameters are monitored. Patient diary cards are often used to track symptoms, which in many cases are an important endpoint.
Depending on the disease model used in a human challenge clinical trial, study participants have to stay in the quarantine after pathogen inoculation for a certain period of time. Such period may vary from couple of days to weeks, as described in a clinical study protocol. This is required not only for the clinical trial data collection purposes, but also to ensure health and safety of the subject, public safety and infection control. Follow-up visits depends on the study protocol but usually the follow-up period is quite short (1-2 months post discharge).
As in all clinical trials – safety of study participants is of most importance. So, is it safe to infect healthy volunteer with a certain pathogen in order to test a new vaccine or antiviral? Challenge studies are designed to limit and minimize risks to participants and are done with strict infection control measures to limit and reduce third-party risks (6). The dose of pathogen to be inoculated is carefully selected, the timing of inoculation is defined in a clinical study protocol. Therefore, with all risk management measures applied, CHIM studies have an excellent safety record.
For example, in the last 30 years of work by the Walter Reed Army Institute of Research conducting CHIM Malaria studies, not one person has been hospitalized due to adverse events related to the study (3). Similarly, in the last 20 years of work by a specialized commercial human challenge study site hVIVO, with over 60 challenge studies conducted and over 3000 healthy volunteers enrolled, these studies have demonstrated an excellent safety profile.
To sum up, human challenge studies has been conducted for many years and provide highly valuable data in clinical development programs. Therefore, such data collected during CHIM studies play an important role in early development of vaccines and anti-infectives. Due to their specifics, challenge studies are conducted only at a limited number of academic centres and a few commercial enterprises, like hVIVO. All such studies require specific clinical research infrastructure, well trained scientists and clinicians, protocols with well-defined inclusion and exclusion criteria, rigorous informed consent processes and careful monitoring and governance (3).
References
Sign up to our emails and follow us on social media to keep up to date with hVIVO.