We look forward to this year’s World Vaccine Congress in Washington DC. With the conference in its 26th year, and the event continuing to grow in size, we believe this to be a great place to connect, talk to and meet with new and old colleagues and contemporaries.
Please do visit us at booth #249 and chat with the team present. Not only will the team be present at the booth, but our Senior Director of Clinical Science, Alex Mann will be presenting in the influenza & respiratory track on April 1 at 14:40 titled “RSV-hMPV-PIV combination vaccine efficacy testing platform”.
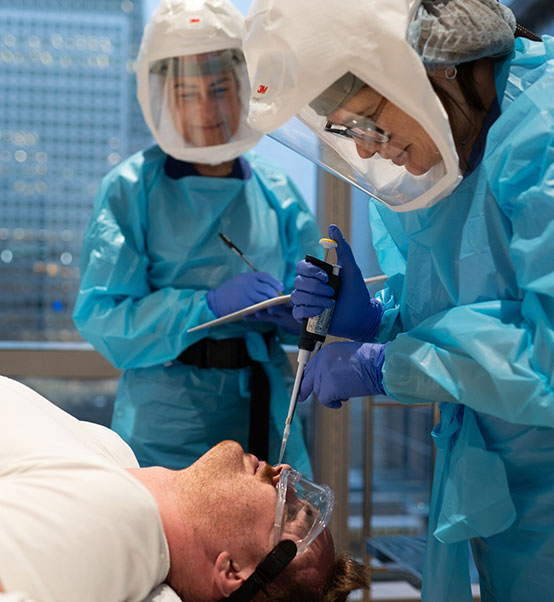
With our team specialising in infectious disease, hVIVO is well positioned to support your vaccine & anti-infective clinical trials targeting a range of infectious diseases, such as:
The breadth of scientific, clinical and research experience ensures that we can support diverse study requirements and deliver reliable, actionable insights for our sponsors.
(RSV-A, RSV-B)
(H3N2,H1N1,H5N1)
(Wuhan, Delta, Omicron)
(HRV-14,HRV-16)
Presented by Alex Mann, Senior Director of Clinical Science
Date: April 1
Time: 14:40
Track: Influenza & Respiratory

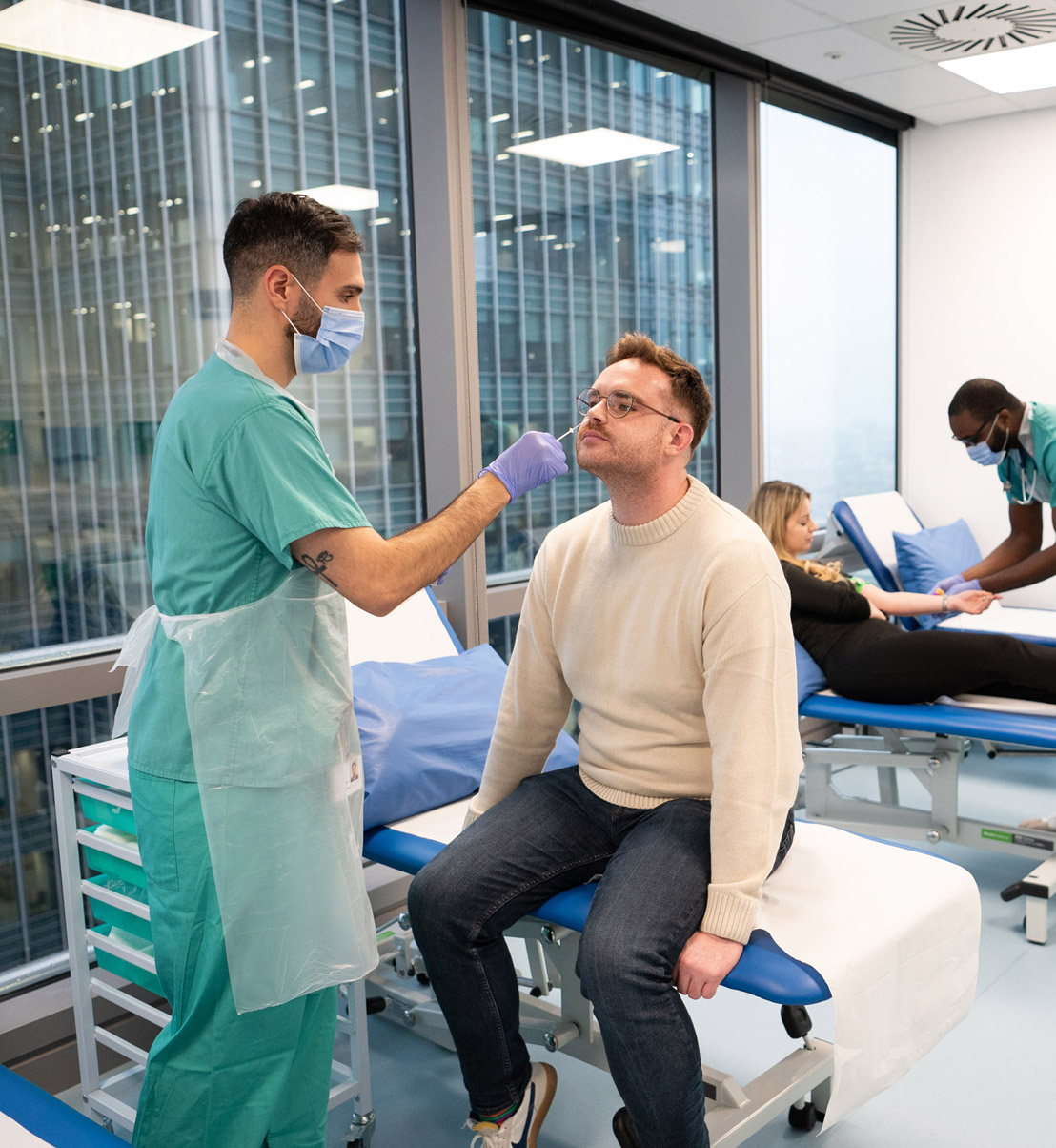
hVIVO supports the full journey from preclinical to Phase III, transforming early Phase I data into clear direction for your product’s next steps. We enable fast success — or fast, informed failure — so you can make smarter decisions sooner.
Our integrated expertise includes:

Building on decades of unrivalled experience, hVIVO is a leading CRO with world-class expertise in delivering human challenge trials, challenge agent manufacture, a unique portfolio of infectious and respiratory disease models, and specialist drug development and clinical consultancy services.

hVIVO has a highly specialised infectious disease and immunology laboratory offering a suite of services to support pre-clinical and drug and vaccine discovery & development.
hVIVO have extended their storage offering to the biotech and pharmaceutical industries with locations in London and Cambridge, offering a dedicated GxP storage facility for biological and clinical materials in secure temperature-controlled conditions.

hVIVO's integrated drug development consultancy supports life science innovators from early planning through clinical execution. With more than 25-years of experience, our team delivers a uniquely combined offering across CMC, non-clinical, clinical pharmacology, regulatory, data management and (translational) laboratory services, across the drug development and market authorisation spectrum.
eBook: Advancing Infectious Disease Vaccine Development
Blog: Advancing research through next-gen HCT models
Webinar: RSV Model
Webinar: hMPV Model
Factsheet: Optimising RSV Sample Workflows
Factsheet: De-Risking Sample Management
Brochure: hVIVO Laboratory Services
Factsheet: Infectious Disease
Full Service Early Phase CRO in Human Challenge Trials
Phase II / III Studies
Sign up to our emails and follow us on social media to keep up to date with hVIVO.